Chemistry Practical Class 11 is an important part of assessment for students. The marks obtained in CBSE Chemistry Class 11 Practical exams are added in the theory marks to prepare the final score of the students in the subject. So, It’s become crucial for students to go through the practicals and activities provided in the Chemistry Lab Manual Class 11. It will help them in scoring high marks in the practical exams. Here, students will find the list of practicals and project works of the Chemistry Lab Manual Class 11 which students have to perform during the academic year.
CBSE Class 11 Chemistry Practical exam consists of 30 marks. Therefore, students must perform Class 11 Chemistry Practicals thoroughly to score high marks. The term 1 Chemistry Practical for Class 11 consist of volumetric analysis for 8 marks, content based experiments for 2 marks , class record and viva for 5 marks. Whereas, CBSE Class 11 Chemistry Term 2 Practical have salt analysis for 8 marks, content based experiments for 2 marks, project work and viva for 5 marks. All these details are provided in the Chemistry Lab Manual Class 11.
CBSE Class 11 Chemistry Practical Experiments
Chemistry is an interesting subject as it gives knowledge about the behaviour of matter, composition, properties, etc. The Class 11 Chemistry practical Exam Syllabus is designed by board in such a way that it evaluates a student’s expertise thoroughly. Students must go through these practicals and Chemistry Lab Manual Class 11 to prepare more efficiently for the exam.
List of Suggested Projects for CBSE Class 11 Chemistry Practical
Scientific investigations involving laboratory testing and collecting information from other sources.
A few suggested Projects
• Checking the bacterial contamination in drinking water by testing sulphide ion.
• Study of the methods of purification of water.
• Testing the hardness, presence of Iron, fluoride, Chloride, etc., depending upon the regional variation in drinking water and study of causes of presence of these ions above permissible limit (if any).
• Investigation of the foaming capacity of different washing soaps and the effect of addition of Sodium Carbonate on it.
• Study the acidity of different samples of tea leaves.
• Determination of the rate of evaporation of different liquids.
• Study the effect of acids and bases on the tensile strength of fibers.
• Study of acidity of fruit and vegetable juices.
CBSE Chemistry Lab Manual Class 11
The list of experiments mentioned in the Chemistry Lab Manual Class 11 are as follow:
EXPERIMENT-1
PREPARATION OF STANDARD SOLUTION OF OXALIC ACID:
AIM:

THEORY:


MATERIAL REQUIRED:

PROCEDURE:


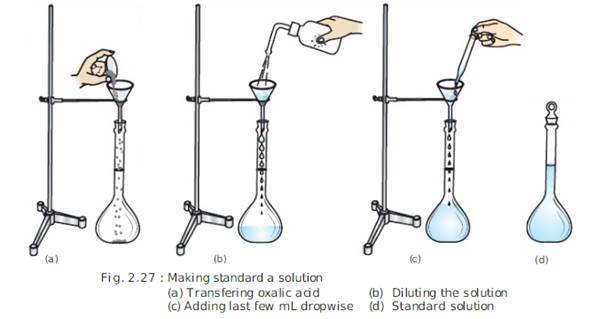
PRECAUTIONS:

EXPERIMENT-2
PURIFICATION OF SAMPLE OF A COMPOUND BY CRYSTALLISATION:
AIM:

THEORY:


MATERIAL REQUIRED:
PROCEDURE:

PRECAUTIONS:

EXPERIMENT-3
DETERMINATION OF MELTING POINT OF AN ORGANIC COMPOUND:
AIM:
THEORY:

MATERIAL REQUIRED:

PROCEDURE:





PRECAUTIONS:

EXPERIMENT-4
DETERMINATION OF BOILING POINT OF AN ORGANIC COMPOUND:
AIM:
THEORY:

MATERIAL REQUIRED:
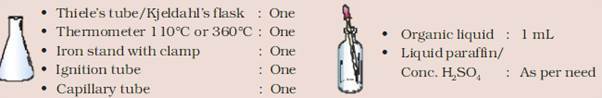
PROCEDURE:

PRECAUTIONS:

EXPERIMENT-5
STUDY OF SHIFT IN EQUILIBRIUM IN THE REACTION OF FERRIC IONS AND THIOCYANATE IONS:
AIM:

THEORY:

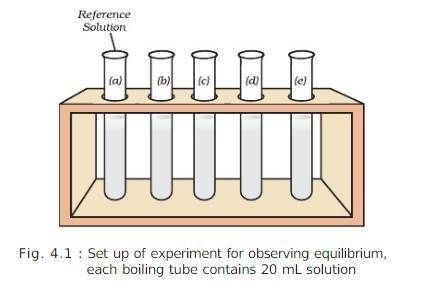
MATERIAL REQUIRED:

PROCEDURE:


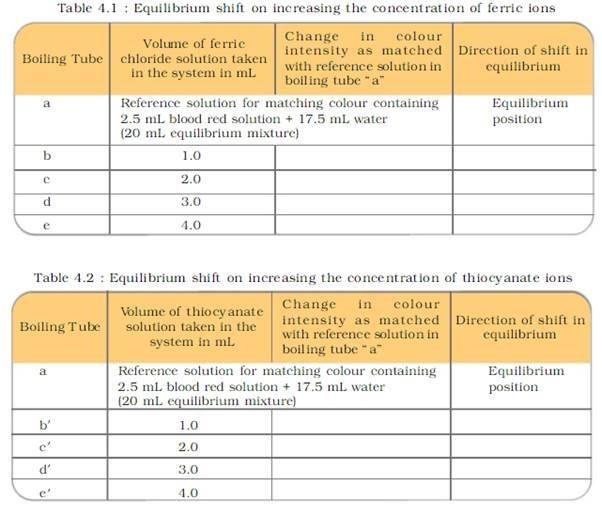
PRECAUTIONS:

EXPERIMENT-6
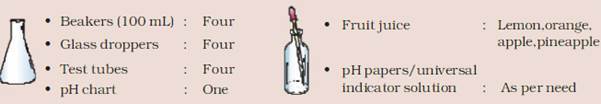
TO DETERMINE THE pH OF SOME FRUIT JUICES:
AIM:
THEORY:


MATERIAL REQUIRED:
PROCEDURE:


RESULT:

PRECAUTIONS:

EXPERIMENT-7
DETERMINATION OF THE CONCENTRATION (STRENGTH) OF A GIVEN SODIUM HYDROXIDE SOLUTION BY TITRATING IT AGAINST A STANDARD SOLUTION OF OXALIC ACID:
AIM:

THEORY:

MATERIAL REQUIRED:

PROCEDURE:




CALCULATION:

RESULT:

PRECAUTIONS:

Stay tuned with BYJU’S to get the latest notifications on CBSE along with CBSE syllabus, sample papers, marking scheme and more.

Osm